luke_stephenson
|
| posted on 15/2/10 at 06:05 PM |

|
|
Any Mechanical Engineers out there?
good evening all,
Maybe someone could help with the following question; the dynamic viscosity of water changes with temperature (known) however does it change with
pressure? i need to know the dynamic viscosity of water @110 celsius 7Bar pressure
cheers
|
|
|
|
|
Bob C
|
| posted on 15/2/10 at 06:21 PM |

|
|
I can't see why it should vary with pressure - 7 bar is not that exotic. I assume you mean kinematic viscosity (visc/density). OK density will
increase ever so slightly with presure.
And if you need it for doing some kind of hydrodynamic simulation other errrors & assumptions will dwarf any physical constant errors!
|
|
|
Nash
|
| posted on 15/2/10 at 07:10 PM |

|
|
quote:
Originally posted by luke_stephenson
good evening all,
Maybe someone could help with the following question; the dynamic viscosity of water changes with temperature (known) however does it change with
pressure? i need to know the dynamic viscosity of water @110 celsius 7Bar pressure
cheers
It's steam at 110 deg C and 7 bar?!?!?!?
...........Neil
It's What You Do Next That Counts.
Build It, Buy It, Drive It:
Southern Kit Car Club
|
|
|
liam.mccaffrey
|
| posted on 15/2/10 at 07:10 PM |

|
|
I was reading an article about this the other day in New Scientist. I think it does change but I can't say for sure.
Build Blog
Build Photo Album
|
|
|
jos
|
| posted on 15/2/10 at 07:16 PM |

|
|
does this help
http://www.engineeringtoolbox.com/water-dynamic-kinematic-viscosit
y-d_596.html
.: Motorsport / motor racing circuit / track wall art Apex Traxs :.
|
|
|
Breaker
|
| posted on 15/2/10 at 07:19 PM |

|
|
quote:
Originally posted by Nash
quote:
Originally posted by luke_stephenson
good evening all,
Maybe someone could help with the following question; the dynamic viscosity of water changes with temperature (known) however does it change with
pressure? i need to know the dynamic viscosity of water @110 celsius 7Bar pressure
cheers
It's steam at 110 deg C and 7 bar?!?!?!?
...........Neil
If the water is pressurized, it will not vaporize at 110 °C. Otherwise lots of cars will have steam cooling instead of water cooling. 
|
|
|
flak monkey
|
| posted on 15/2/10 at 07:20 PM |

|
|
quote:
Originally posted by Nash
It's steam at 110 deg C and 7 bar?!?!?!?
...........Neil
Nope still a liquid I think at that pressure and temperature.
Sera
http://www.motosera.com
|
|
|
will121
|
| posted on 15/2/10 at 07:35 PM |

|
|
quote:
Originally posted by flak monkey
quote:
Originally posted by Nash
It's steam at 110 deg C and 7 bar?!?!?!?
...........Neil
Nope still a liquid I think at that pressure and temperature.
definately still be a liquid, cant get my steam tables out of the loft be covered in 20years of dust, but we run high temp Hot water heating systems
upto 150degC @10bar,
|
|
|
luke_stephenson
|
| posted on 15/2/10 at 08:26 PM |

|
|
with ref to engineers toolbox, thankyou, id already checked there tho. v.good site!
afraid its definately still in the liquid state, saturation temp @ 7 bar is 165celsius.
ok so this is my theory, as shown in the engineers toolbox and other sites the dynamic viscosity which is different to kinematic viscosity varies with
temperature @ atmospheric pressure. however the chart only goes to 100c obviously as the water will boil. and it is shown that as the water nears
boiling point the dynamic viscosity lowers.
all well and good, but the 7bar pressure would effectively have the water in a totally different state as it would not be near boiling point. its got
me well stumped.
unfortunately i need it to get a reliable reynolds number to allow accurate heat transfer calcs and more importantly accurate scaling of the proposed
heat exchanger.
im well and truely stuck!
thanks for all the input so far
|
|
|
Nash
|
| posted on 15/2/10 at 08:49 PM |

|
|
quote:
Originally posted by will121
quote:
Originally posted by flak monkey
quote:
Originally posted by Nash
It's steam at 110 deg C and 7 bar?!?!?!?
...........Neil
Nope still a liquid I think at that pressure and temperature.
definately still be a liquid, cant get my steam tables out of the loft be covered in 20years of dust, but we run high temp Hot water heating systems
upto 150degC @10bar,
I thought 110 was a turning point?! So I found this and I was wide of the mark
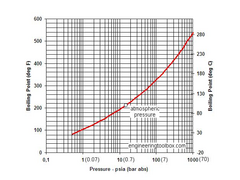 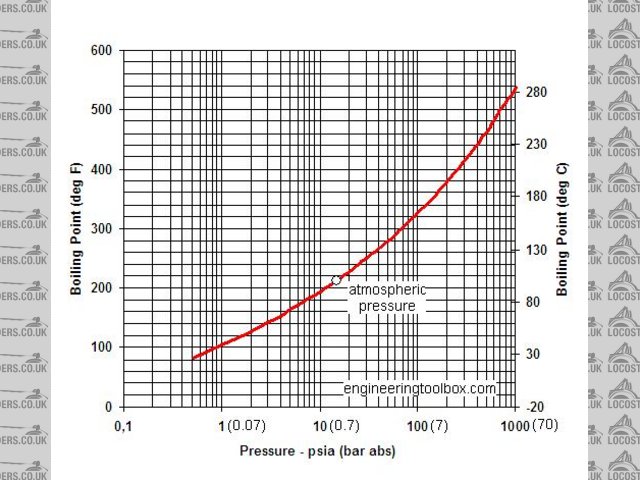
Steam tables
It is about 150 by the look of things
.......Neil
It's What You Do Next That Counts.
Build It, Buy It, Drive It:
Southern Kit Car Club
|
|
|
Ivan
|
| posted on 15/2/10 at 08:51 PM |

|
|
This is where I normally pick up the phone and phone the Varsity and ask to speak to the relevant Prof - normally they are very happy to help if you
approach them civilly and explain your problem.
|
|
|













